The MRC Histology and Morphometry Core has recently enhanced its protocols for the sectioning, imaging and analysis of skeletal muscle morphology. If you are interested in these services, please read follow these tips for preparing and delivering your muscle sample to ensure that you get the best images to answer your scientific question.
Preparation of Muscle Samples
- Decide whether you will use fresh frozen or fixed samples. Fresh frozen samples retain the best structural morphology and are recommended for structural analyses such as fiber areas and extracellular matrix area fraction. Fixed samples may work better with some antibodies or in cases of severe pathology where structures are fragile. If you are unsure which is best, contact the technical staff for guidance.
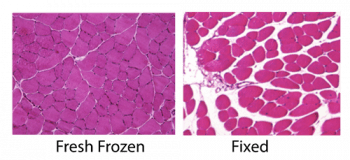
- Prepare your muscle according to the guide below and deliver to Core for sectioning.
Fresh Frozen
- Dissect your muscle of interest, place in PBS moistened gauze.
- If you are unsure which muscle to use for analysis or how to dissect said muscle, please contact the Core technical staff for assistance.
- Facilities are available in the Core to perform dissections on site.
- Deliver to the Core as quickly as possible for embedding and freezing. Avoid soaking the muscle as this can cause fiber swelling.
Fixed
- Dissect your muscle of interest, place in neutral buffered formalin for 24 hours.
- Sink the muscle in 30% sucrose for an additional 24 hours.
- Deliver the muscle in sucrose to the Core for embedding and freezing.
Resources for Histological Imaging and Analysis
Once your muscle has been sectioned, the Core can provide assistance with routine stains to evaluate muscle morphology and pathology (examples below). For more information, contact the Core technical staff.

Core Contacts

Crystal Idleburg, HT(ASCP)
Senior Research Tech/ Histology
- Phone: 314-747-6034
- Email: idleburg_c@nospam.wustl.edu

Gretchen Meyer, PhD
Physical Therapy & Neurology
- Phone: 314-286-1425
- Email: meyerg@nospam.wustl.edu
Focus: changes at the molecular and cellular level in skeletal muscle and how they affect muscle structure and function.